What we do
Our Capabilities
We offer comprehensive information technology professional services for all business needs including data integration, custom application development, commercial-off-the-shelf (COTS) product integration, data reporting & analytics, and application & information security.
Digital Transformation
We modernize legacy processes using Business Process Reengineering (BPR), identifying pain points, designing innovative solutions, and refining them for improved efficiency and customer satisfaction.
DevSecOps
We accelerate software delivery using automated DevSecOps pipelines, ensuring reliable releases, reduced errors, and cost-efficient scalability. We build secure software by embedding secure coding practices and monitoring system performance to optimize operations and enhance security.
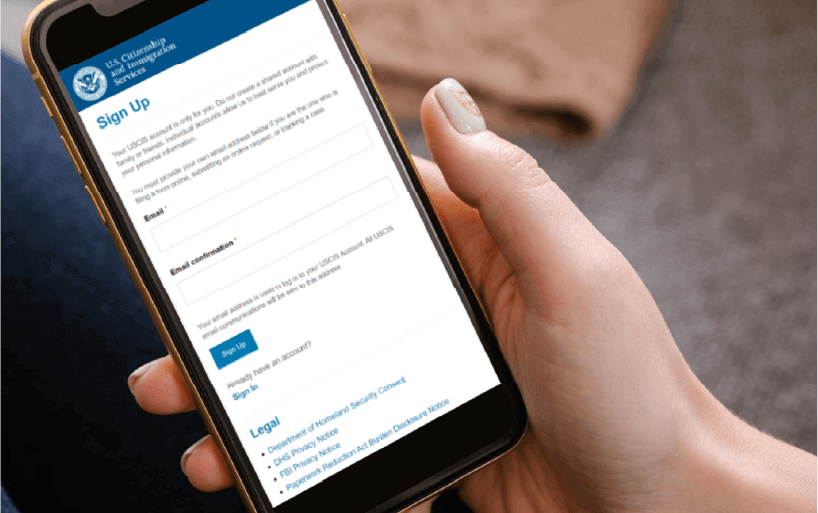
Information Security
We provide enterprise authentication and authorization solutions, using multi-factor authentication and fine-grained access controls to enhance security. Embracing Zero Trust, we implement continuous verification and least-privilege access to proactively detect and respond to threats.
Data Science & AI/ML
We transform big data into actionable insights using tools like Databricks, Apache Spark, Kafka, and Airflow for efficient processing and real-time pipelines. Our AI/ML and data visualization expertise helps clients make smarter decisions and communicate compelling data-driven stories.
Who we work with
We are proud to support a diverse array of federal government customers who value innovation, creativity, and excellence. Our clients appreciate our commitment to delivering high-quality services that enhance operational efficiency and drive mission success.
Our Clients









Our Partners